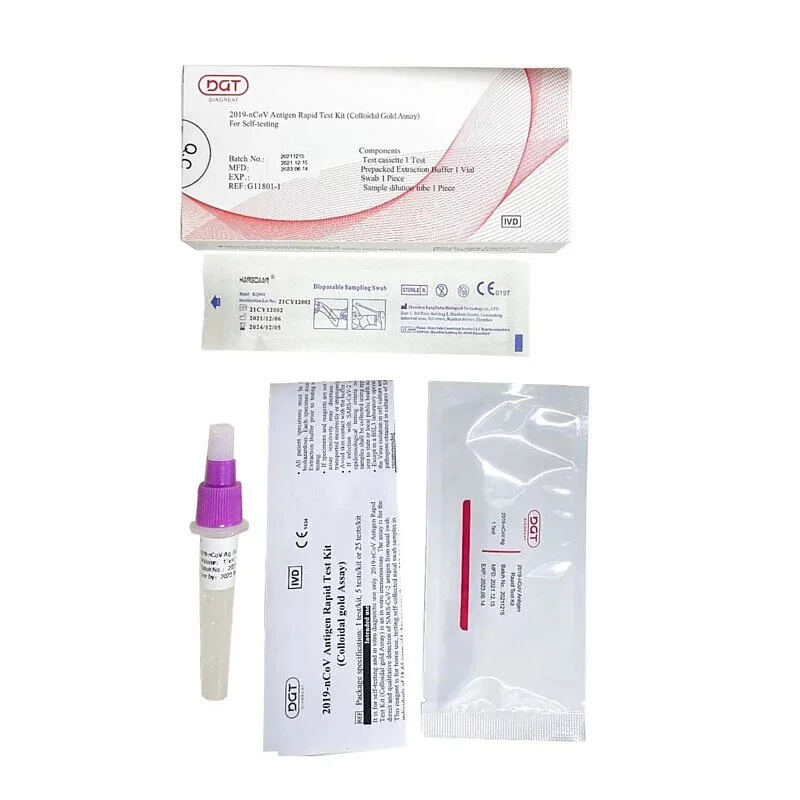
DIAGREAT Covid-19 抗原快速檢測試劑盒
- #COVID19快速測試劑
- 居家自行檢測劑
- 用法簡單,測試只需15分鐘
疫情持續蔓延,有陣時都唔知自己係真係中左定係普通感冒
亦或是明明冇事去左檢測中心一個唔小心就中左啦‼️ 病毒真係無所不在
除左做好自己嘅衛生保護之外都有其他辦法快速保護到自己同屋企人啦
- 最短約15分鐘有測試結果
- 能在家簡單自我檢測
- 準確度達96.44%
- CE&KKM&MDA多個國製認證
- Nasal 鼻子液測試
適用於:
- #新型冠狀病毒感染
- #變種病毒
產地:中國
有效期:約1年,儲存溫度不能高於40度
🔥使用方法🔥
樣本收集,在鼻腔順時針及 逆時針轉動。
準備樣本,把 鼻腔棒放到抗原提取管當中。
進行測試,垂直倒轉試管,將兩滴測試樣本加入樣本孔中。
開始計時,並等待15分鐘。
等待測試結果,並對照結果,如超過20分鐘,結果則不再有效。
購前注意
* 此產品非正式醫療用品,測試結果只供參考
* 檢測結果不適用於強檢報告/其他相關政府文件申請/旅遊申請
*任何出現症狀者,建議及早檢測及就醫
* 如結果呈陽性,請及早帶同結果至醫管局認可醫療機構求醫
* 圖片效果只供參考,外盒會因不同國家版本而有所差異, 內容則不變
2019-nCoV Antigen Rapid Test Kit
In 2020, Coronavirus (COVID-19) is raging around the world. In February, Diagreat successfully developed the Coronavirus Antigen kit, 2019-nCoV Antigen Rapid Test Kit, which is based on the Colloidal Gold Assay.
Intended use
The rapid test kit is used for qualitative determination of novel coronavirus (SARS-CcV-2) antigen in human nasal swab and saliva samples in vitro.. This kit is offered to clinical laboratories and healthcare workers for point-of-care testing, and not for at home testing, in compliance with Section IV.D. of the FDA’s Policy for COVID-19 Diagnostic Test.
Principle
After an appropriate amount of sample is added to the detection well, the sample moves under the action of the capillary. The new coronavirus antigen in the sample will combine with colloidal gold-labeled new coronavirus N protein antibody to form a colloidal gold-antigen-antibody complex. The immune complex product is then chromatographed along the nitrocellulose membrane to the detection area (T), binds to the pre-coated N protein monoclonal antibody, and forms a purple line, indicating that the new coronavirus antigen is positive.
The quality control antibody-labeled colloidal gold particles are chromatographed to the quality control area (C) and combined with the pre-coated anti-quality control antibody to form a purple C line, indicating that the test is effective. If the QC line does not appear, the test result is invalid.
Applicable Department
Emergency Department
ICU
Pneumology Department
Cardio-Pulmonary Function Department